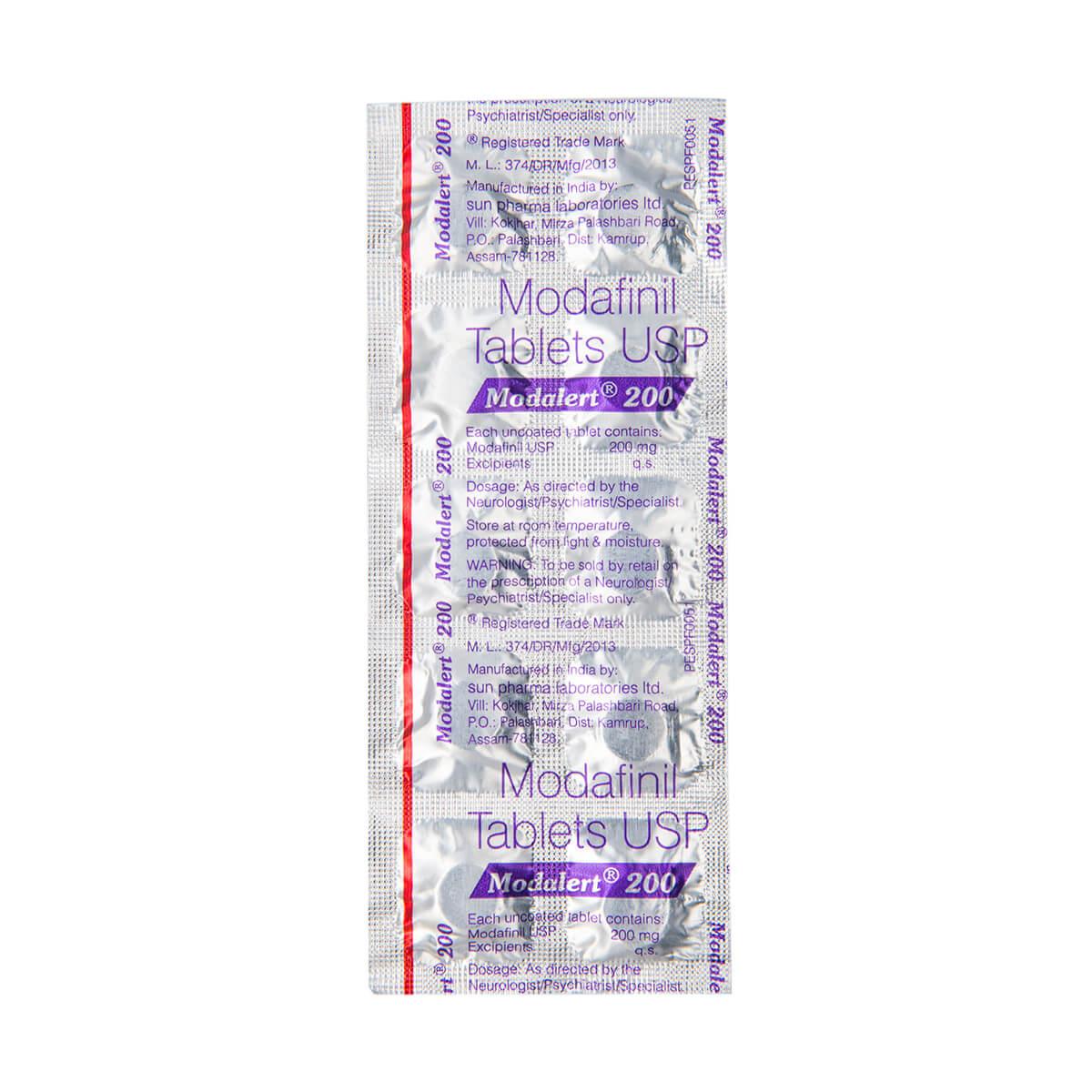
Modafinil is a wakefulness-promoting agent and a eugeroic drug that is used to treat various sleep disorders such as narcolepsy, obstructive sleep apnea/hypopnea syndrome, and shift work sleep disorder. It is also used off-label to improve cognitive function, increase focus and alertness, and enhance productivity. Modafinil belongs to a class of drugs known as analeptics and works by stimulating certain areas of the brain that are responsible for wakefulness and alertness.
The chemical name for Modafinil is 2-[(diphenylmethyl)sulfinyl]acetamide, and it has a molecular formula of C15H15NO2S. Modafinil is a white to off-white crystalline powder that is insoluble in water and slightly soluble in ethanol and methanol. It is available in tablet form for oral administration in various strengths, including 100mg, 200mg, and 400mg.
Modafinil is metabolized by the liver and has a half-life of approximately 12-15 hours. It is eliminated from the body primarily through the kidneys, with approximately 90% of the drug being excreted in the urine and the remainder in the feces.
Modafinil is a Schedule IV controlled substance in the United States due to its potential for abuse and dependence. It should only be used under the supervision of a qualified healthcare provider and should not be used by individuals with a history of drug abuse or addiction.
Modafinil has been shown to improve cognitive function in healthy individuals and to enhance performance on tasks that require sustained attention and vigilance. It has also been shown to improve memory and learning in animals and may have similar effects in humans. Modafinil is thought to work by increasing the release of certain neurotransmitters in the brain, including dopamine, norepinephrine, and histamine.
Modafinil has a low incidence of side effects, with the most common being headache, nausea, and nervousness. More serious side effects are rare but may include skin rash, fever, and liver damage. Modafinil may interact with other medications, including hormonal contraceptives, and should not be used by individuals with liver or kidney disease.
In conclusion, Modafinil is a wakefulness-promoting agent that is used to treat various sleep disorders and to enhance cognitive function in healthy individuals. It works by stimulating certain areas of the brain that are responsible for wakefulness and alertness and has a low incidence of side effects. Modafinil should only be used under the supervision of a qualified healthcare provider and should not be used by individuals with a history of drug abuse or addiction.




Reviews
There are no reviews yet.